One patent, many applications

When Dr. Parminder Vig meets people living with neurodegenerative diseases such as cerebellar ataxias, Alzheimer's and Parkinson's disease, he senses a call to action.
“When you meet patients with these conditions, you wonder why you can't do something about neurological diseases,” said Vig, a professor of neurology and biochemistry at UMMC.
Vig is certainly doing something: he studies the molecular cause of the disease spinocerebellar ataxia (SCA) in order to develop therapies. Now, he has a patent that could help these patients.
The patent is for a technique to deliver bioactive peptides to the brain and spinal cord using an elastin-like polypeptide (ELP) and a cell-penetrating peptide. Vig's co-inventors are Dr. Drazen Raucher, professor of biochemistry and Dr. Scoty Hearst, a former graduate student.
Ataxia refers to uncoordinated movements like walking or balance problems caused by neurological changes. Vig says that a drunken stupor is a type of temporary ataxia. Neurological disorders also cause permanent ataxias such as SCA, a group of genetic conditions that affect cells in the spinal cord and the cerebellum, the brain region that controls body movements.
Vig, Raucher and Hearst used the disease SCA1 to develop the patent. In SCA1, a mutation causes the protein ataxin-1 to gather inside of cerebellar neurons called Purkinje cells. Ataxin-1 builds up to toxic levels and kills the cells. As the cells die, the patient loses motor control and the disease becomes fatal.
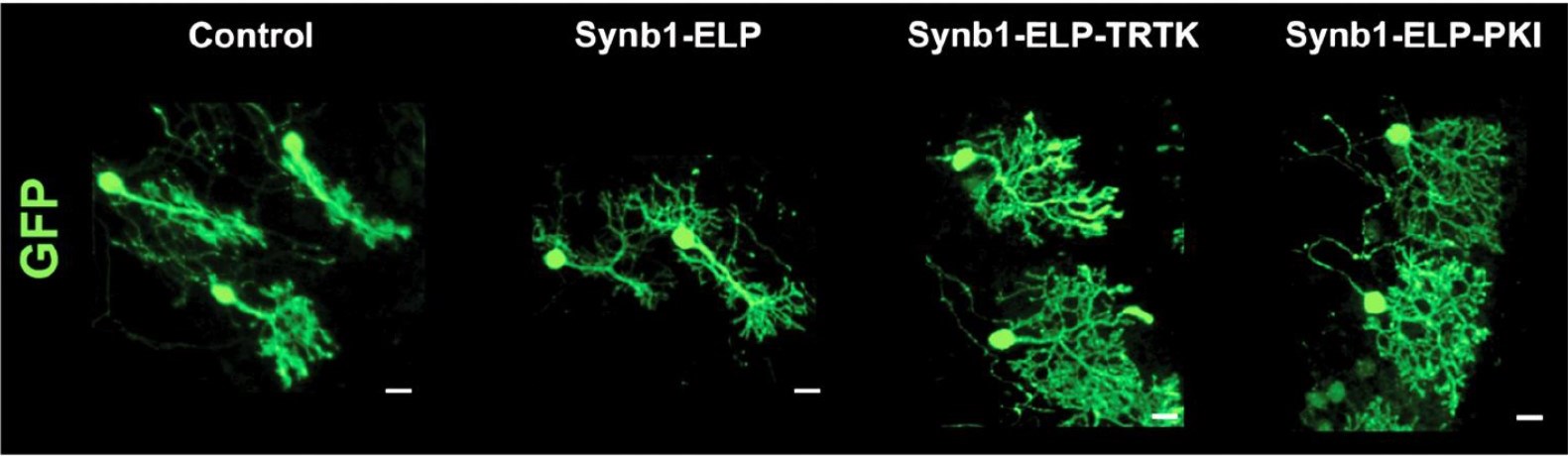
“In neurological diseases, we don't see symptoms unless fifty percent of the cells are gone,” Vig said. “Our goal is to target symptoms before they start by delaying cell degeneration.”
Vig's drug candidate, a small peptide, works by slowing ataxin-1 buildup. However, the drug's short half-life means the body degrades and removes it before there is a lasting therapeutic effect.
Raucher studies chemotherapy delivery using ELP, an engineered amino acid chain that can be attached to therapeutics. ELP is water-soluble below human body temperature, but aggregates when warmed up a few degrees. This quality makes ELP useful for targeted drug delivery that protects the therapy from degradation and helps eliminate side effects, Raucher says.
However, the brain presents a challenge.
“If you want to deliver drugs to the brain, then the blood-brain barrier can be a problem,” Raucher said.
The blood-brain barrier is made of tightly joined cells that prevent most foreign agents from passing through, including ELP. One way to get past the wall is to drill a hole. So, the researchers added a molecular drill bit: a cell-penetrating peptide that breaks through the blood-brain barrier and allows the ELP and drug to reach the cerebellum.
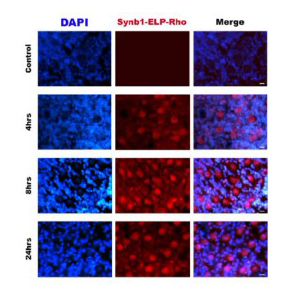
Using a mouse model of SCA1, they were able to show that the drug aggregates in the heated cerebellum. The experiments are the first to show that ELP-based therapeutics can be targeted to the brain and nervous system.
Furthermore, the SCA1 mice that received the treatment had better balance and motor coordination than controls, suggesting that the treatments could work for people as well.
The patent covers not only the molecules, but also how they are administered to the patient.
First, a laser heats the brain over a series of cycles instead of the typical approach used for ELP-based therapeutics. The laser allows for greater skull penetration and focuses the heat in a smaller area.
The delivery route is another innovation. Vig, Raucher and Hearst used olfactory pathway to the brain by administering drug up the nose, instead of intravenously.
“This allows the drug to reach the target faster and more efficiently,” Raucher said.
Although they developed the patent using SCA1 as a model disease, Vig says that the patent's broad scope makes the technology versatile.
“This patent has so many applications by taking a top-down approach,” Vig said, that allow its use in human and veterinary medicine, according to the patent.
“It is a platform that we can apply to different diseases,” Raucher said.
For example, the technique could be a treatment option for other neurodegenerative conditions such as Alzheimer's and Parkinson's diseases. Major depression, brain tumors and brain and spinal cord injuries could be treated as well.

“It's a highly innovative method for overcoming the blood brain barrier and delivering therapies to targeted regions,” said Dr. Tom Mosley, director of the MIND Center at UMMC. “It's an exciting advance and will substantially expand our ability to test new treatments for neurodegenerative diseases like Alzheimer's.”
This patent has not been tested on human patients and may not be for several years. However, other labs are conducting clinical trials for a diabetes treatment using ELP. Vig and Raucher say that this work provides evidence that their own ELP method can reach the trial stage and improve the lives of people with SCA and other conditions.
“The patient's focus is to get better and we are going to continue our work to make that happen,” Vig said.
The research used to develop the patent was funded by the National Institutes of Health.


