Study Activation
Main Content
Study Activation Home
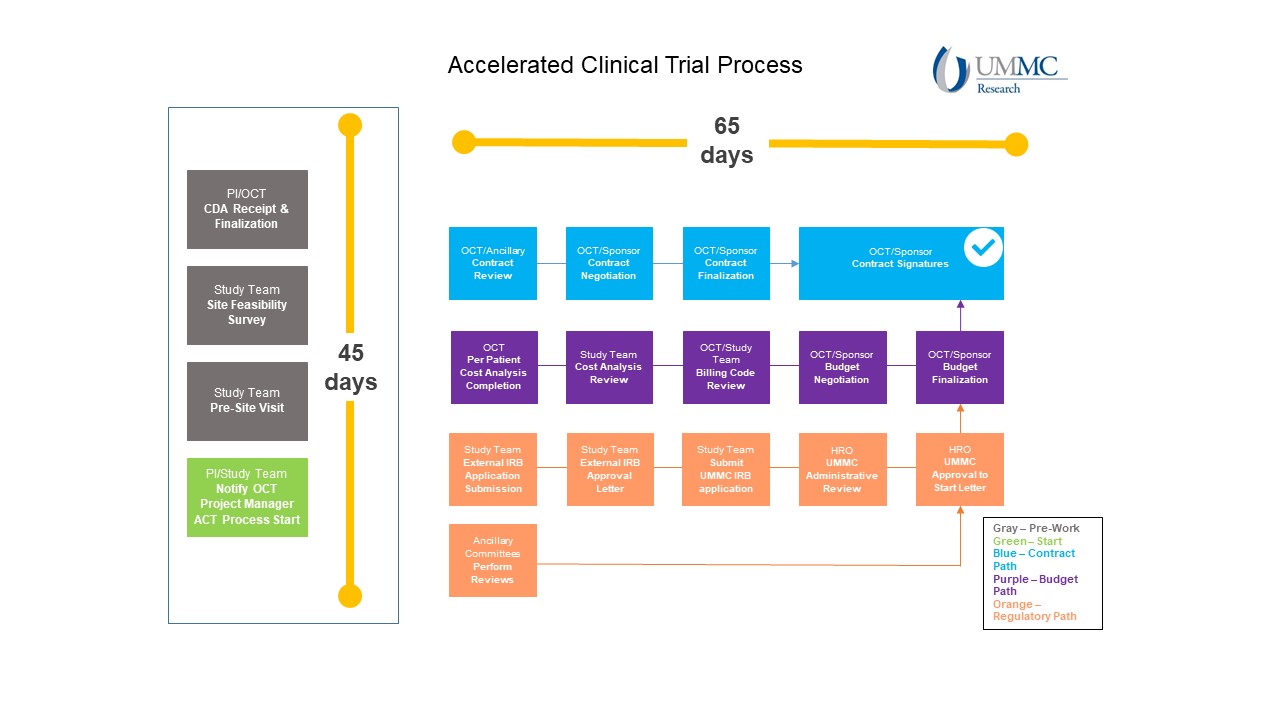
UMMC clinical research study activation is governed by the Activation of Clinical Trials (ACT) process. During this process, the contract and budget are negotiated finalized, UMMC Institutional Review Board (IRB) approval is received, and the clinical research study agreement is executed.
UMMC's goal is to complete the ACT process within 65 calendar days. Once this process is complete, the study and site information should be added to clinicaltrials.gov by the responsible party, as applicable.
The graphic below provides an overview of the activation process.
Use the left-hand navigation pane to learn more about each step in the study activation process.


