Cancer Center and Research Institute
The Cancer Center and Research Institute unites physicians, nurses and researchers from the University of Mississippi Medical Center and the University of Mississippi in the quest to stop cancer through:
- Interdisciplinary world class cancer prevention, detection and treatment.
- Research to better prevent, detect and treat cancer.
- Education for Mississippi residents and the medical caregivers who one day will provide their cancer care.
Adult and pediatric physicians see more than 2,000 new patients annually and follow more than 5,800 survivors. Cancer teams see many people with rare or complex care needs.
Keep up with Cancer Center and Research Institute news by connecting with us through social media. CCRI is active on Facebook, Instagram, X and LinkedIn.
Mississippians deserve world-class cancer care at home.
UMMC is leading a bold effort to save more lives by building a new, 250,000 square-foot cancer center, a state-of-the-art facility that unites care, research and clinical trials in one place.
Mississippians deserve world-class cancer care at home.
UMMC is leading a bold effort to save more lives by building a new, 250,000 square-foot cancer center, a state-of-the-art facility that unites care, research and clinical trials in one place.
Learn more and donate to the campaign.

Featured News

Trustmark gives $2M gift to campaign for UMMC Cancer Center and Research Institute
Wednesday, May 26, 2027
Trustmark’s donation will help UMMC build a new home for cancer care and research, supporting the Medical Center’s work to expand treatment options for patients across Mississippi. Read More

Front and Center: Marlene Rios
Monday, May 11, 2026
Before patients meet their care team or hear a treatment plan, they meet someone who instantly puts them at ease. For CCRI, that reassuring presence is often Marlene Rios. Read More
Latest News

McRae Foundation’s generosity continues with significant gift to UMMC cancer center campaign
Published on Monday, May 4, 2026
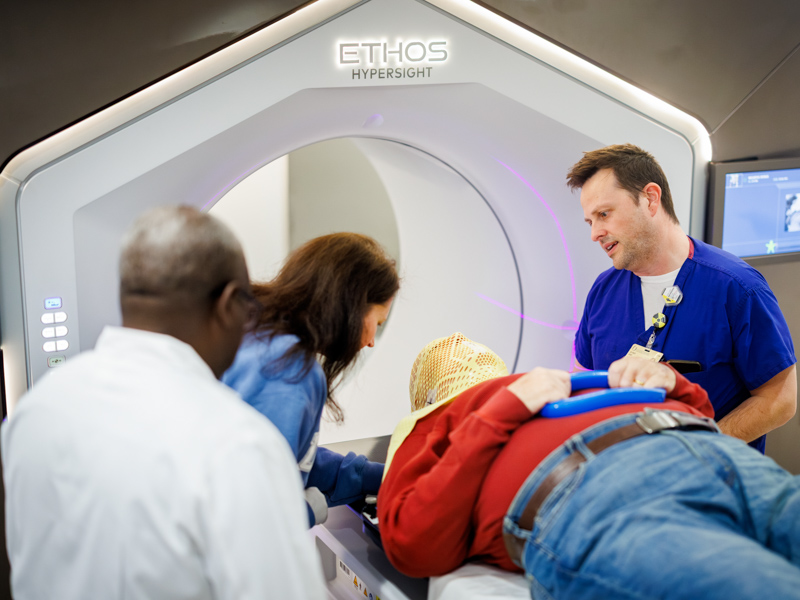
New state-of-the-art radiation oncology systems offer advanced cancer care at UMMC
Published on Monday, April 20, 2026
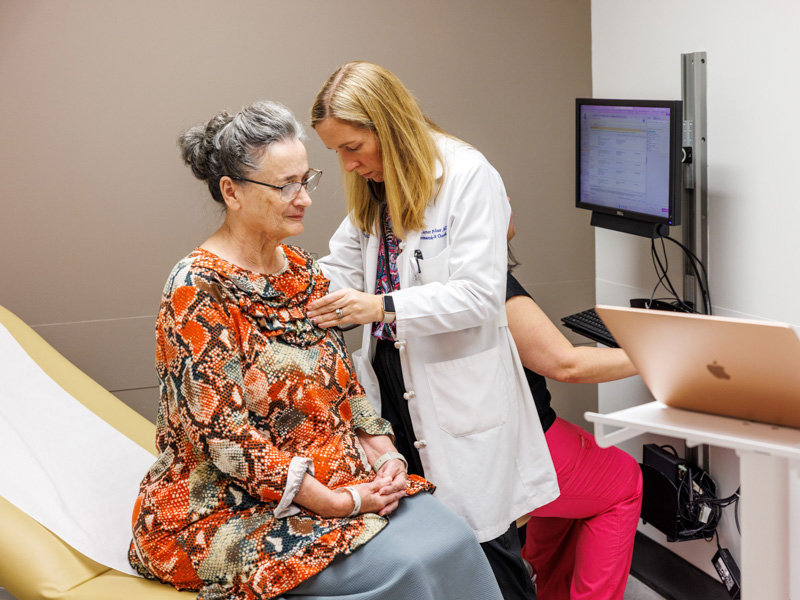
BiTE immunotherapy offers new hope for patients with aggressive cancers
Published on Monday, April 6, 2026

UMMC Cancer Center and Research Institute announces public phase of $125M campaign
Published on Monday, March 30, 2026

Transforming care through laughter: Olivers make $500K gift to CCRI
Published on Monday, March 16, 2026

Bower Foundation makes $10M gift to campaign for UMMC Cancer Center and Research Institute
Published on Monday, March 9, 2026

Compretta earns 2026 SEC Faculty Achievement Award
Published on Monday, March 9, 2026
Treatment
Interdisciplinary teams meet regularly to review each new patient’s case and recommend the best treatment for each individual. Doctors here see many unusual and complex cases.
Health Care
Education
The Cancer Center and Research Institute offers the state’s only residency and fellowship programs for professionals seeking a career in cancer care.
Students and Trainees
Research
Researchers and physicians at UMMC and at the University of Mississippi are working together to seek new and better ways to prevent, detect and treat cancer.
Research Overview