Clinical trial participants gain ‘greater awareness’ of health
Researchers at the University of Mississippi Medical Center are committed to finding new and innovative ways to prevent and treat the diseases that affect our daily lives.
The ultimate goal: to improve the well-being of all people.
Clinical studies facilitate the advancement of medicine, but that progress cannot happen without research partners – members of the public who volunteer for said studies.
Now, UMMC has a new tool that can help people learn more about clinical trials at the Medical Center and how they can become involved.
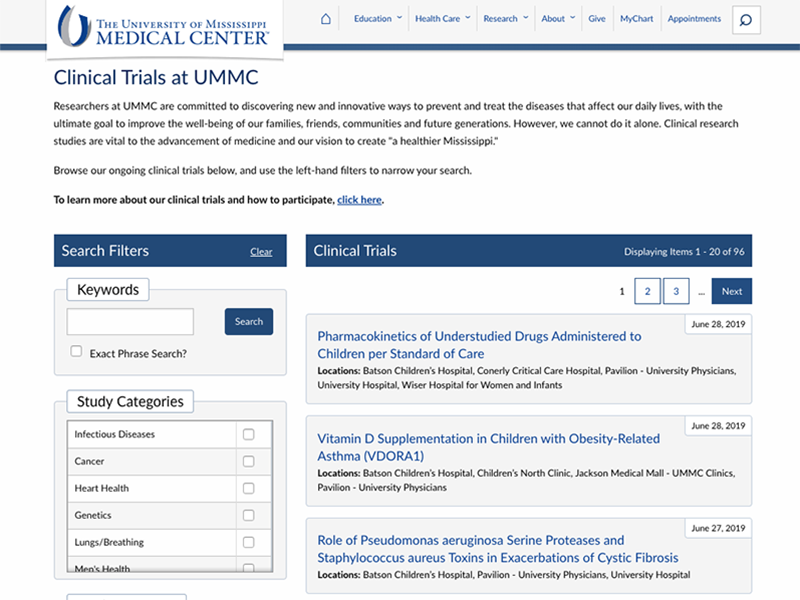
Introduced in June, the “Find a Trial” webpage includes information about ongoing studies at UMMC. The tool contains information on experimental studies of new drugs and procedures as well as observational studies that are recruiting participants.
By clicking a trial name, users can view more detailed information about the purpose of the study; participant inclusion and exclusion criteria; and contact information for the research team.
Whitney Bondurant, director of UMMC’s new Office of Clinical Trials, said the interface improves earlier efforts to bring visibility to ongoing clinical trials.

“We wanted to make it more transparent, engaging and user-friendly,” Bondurant said.
The site’s design was inspired by clinicaltrials.gov, the National Institutes of Health’s database of clinical studies underway around the world. Users can filter through the list to find trials related to particular health conditions, recruitment criteria, study phase and locations where the study visits will occur.
As of this writing, UMMC’s page lists 96 trials enrolling participants. They include multiple studies associated with cancers, cardiovascular diseases, neurological conditions and other diseases in both children and adults.
Bondurant said the page will be updated regularly as new studies are approved by UMMC’s Institutional Review Board and move to the recruitment phase.
The page is the latest in a series of efforts to increase public visibility and knowledge surrounding clinical research at UMMC.
In the broader community, Bondurant said, “People are unaware of how they can learn more about enrollment or participation in clinical trials.”
A global 2018 study found that 85 percent of people view clinical research as important, but nearly 60 percent could not name a place that conducted this kind of research.
People also can submit a general interest form stating their interest in receiving more information about future trials. This builds a pool of potential participants for future studies. People who aren’t current UMMC patients can request information and participate in studies as well.
Information submitted through these tools is protected by patient privacy laws and there is no obligation to participate if contacted by UMMC about an upcoming trial.
“One of the biggest benefits people report from participating in clinical research is a greater personal awareness of their health,” Bondurant said. “This can have a broader public health impact than access to experimental treatments alone.”
The above article appears in CONSULT, UMMC’s monthly e-newsletter sharing news about cutting-edge clinical and health science education advances and innovative biomedical research at the Medical Center and giving you tips and suggestions on how you and the people you love can live a healthier life. Click here and enter your email address to receive CONSULT free of charge. You may cancel at any time.



