Academic Effectiveness
- Office of Academic Affairs Home
-
About Academic Affairs
- About Academic Affairs
- Academic Affiliations
- Academic Effectiveness
- Academic Engagement
- Academic Excellence
- For Prospective Students
-
For Students
-
Student Affairs
- Office of Student Affairs
- Student Organizations
- Contact Student Affairs
- Student Life Calendar
- Associated Student Body
-
Student Accounting
- Student Accounting Home
- Tuition Information
- UMMC Waivers
- Tax Information
- Student Insurance
- Contact Us
- Student Counseling Services
- TELUS Health Assistance Program
- Student Benevolence Fund
- Police and Security and Transportation
- Suggestions for the Student Union
- Student Complaints
- Title IX and Sexual Misconduct
- Student Information Systems
- Student Success
- Academic Outreach Programs
- Academic Engagement
- Interprofessional Education
- GivePulse and Service Learning
- Academic Innovation
- Enrollment Management
- Off Campus Housing
- International Services
- Student Accounting and Insurance
- Student Financial Services
- Commencement
-
Student Affairs
- For Faculty
- Resources
- Museum of Medical History
Academic Assessment
Academic Assessment is part of the Office of Academic Compliance, Effectiveness, and Research (ACER).
Our services
Our assessment staff:
- educate and provide best-practice resources for tracking the outcomes and effectiveness of institutional, instructional programs, and administrative-educational support services,
- uphold the highest standards for continuous quality improvement in alignment with UMMC's academic mission,
- coordinate ongoing, systematic planning and reporting efforts by managing institutional assessment procedures,
- assist with demonstrating compliance for accreditation and
- assess findings and data used for targeted performance enhancement and leveraged to promote excellence in student-centered training and service initiatives.
Our framework
The assessment framework supports aligning unit/program objectives with UMMC's strategic goals. It provides a structured approach for gauging unit/program impact in the delivery of service or instruction. Mission statements, outcomes, means of assessment, success criteria, and results summaries are annually evaluated to support decision-making. Improvement over time is examined by reflecting on data from previous cycles and appraising actions for their ability to impact outcome metrics positively. Current data is then used to inform enhancement initiatives' implementation and plan future outcome assessments.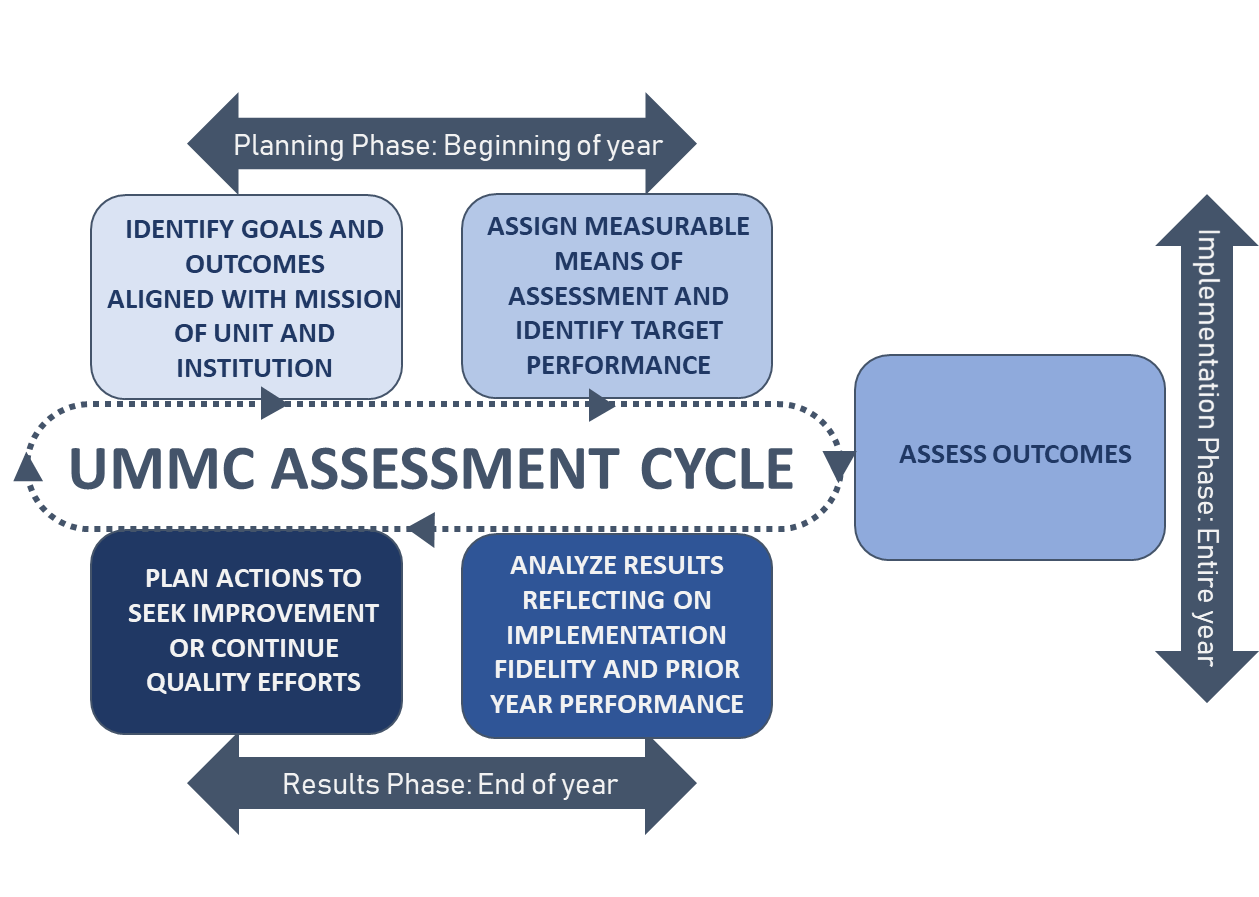
The schematic represents significant elements of the assessment framework used at UMMC to capture assessment data and demonstrate the effectiveness of unit/program outcomes.
Assessment cycle
The assessment process at the University of Mississippi Medical Center (UMMC) is an ongoing, research-based, systematic process that embodies training, a framework for data collection, and internal report review to examine and document effectiveness across education-related support services and degree programs on an annual basis to seek improvement of desired outcomes.
The assessment process is conducted over the fiscal year (July 1 through June 30). It includes notifications, training, and submission deadlines essential for conducting and managing the review of unit/program assessment plans and effectiveness reports.
UMMC faculty and staff may access more information by searching "Assessment" on the UMMC Intranet. For more information on the assessment process, email the Assessment or contact the ACER team.


